Newswise — Losing a large amount of soft tissue from an injury or cancer may require reconstructive surgery. These surgeries typically rely on a structural framework that holds cells or tissues together made from hydrogels or other biomaterials that provide support for new blood vessels to grow. But when these frameworks are made from bulk hydrogels, they have several limitations that can result in slow and disorganized blood vessel growth, leading to poor patient outcomes.
To overcome these limitations, researchers at The Pennsylvania State University have developed a new synergistic approach that combines a new framework (scaffold) made from granular hydrogels with a surgical technique called micropuncture. Their preclinical method, published in the journal Small, could rapidly grow organized blood vessels in live rats.
“The researchers’ hybrid approach to revascularization represents a significant advance in the field of tissue engineering and regeneration. This technique could be applied to a variety of blood-vessel conditions found in cardiovascular disease, such as coronary heart disease, the most common type of heart disease in the U.S.,” said Tuba Fehr, Ph.D., program director in the NIBIB Division of Discovery Science and Technology.
Hybrid approach for revascularization
Surgical micropuncture is a preclinical procedure developed by co-senior study author Dino Ravnic, D.O., the Huck Chair in Regenerative Medicine and Surgical Sciences at Penn State College of Medicine. The procedure involves making tiny, precise cuts in the blood vessels, which accelerates vascularization. While new blood vessels may grow rapidly, past studies show they do not form well-organized patterns when combined with bulk hydrogel scaffolds.
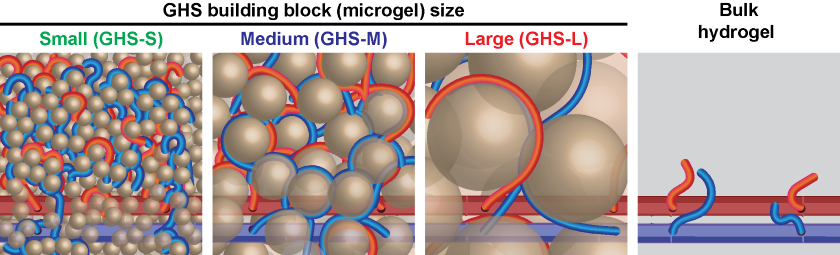
To encourage blood vessels to grow in a more orderly manner, senior study author and principal investigator Amir Sheikhi, Ph.D., Dorothy Foehr Huck and J. Lloyd Huck Early Career Chair in Biomaterials and Regenerative Engineering at The Pennsylvania State University, developed protein-based granular hydrogel scaffolds. They are made from hydrogel particles (microgels) that are one-thousand times larger than the polymer chains/nanoparticles found in bulk hydrogel scaffolds. The nanoparticles create smaller pores that have limited blood vessel organization and interconnection in bulk hydrogels.
In contrast, microgels create larger pores that enable rapid cell ingrowth and vascularization. To guide the architecture of blood vessels, the microgels were created in different sizes (small, medium, and large).
“To form the patterns for blood vessels, we adjusted the empty spaces (pores) among microgels by changing their size. The smaller the microgels, the smaller the median pore diameter, and the smaller the intercapillary distance when blood vessels form,” said Sheikhi.
Evaluation in animal models
The researchers then evaluated their hybrid approach in live rats. Before implanting their granular hydrogel scaffolds into a recipient, the researchers performed vascular micropuncture to accelerate the formation of new blood vessels. The perforations are made with microneedles to ensure there are no blood clots or significant bleeding. Then, after implanting the granular scaffolds, the researchers compared their hybrid approach to rats that received the bulk hydrogel scaffolds with or without micropuncture.
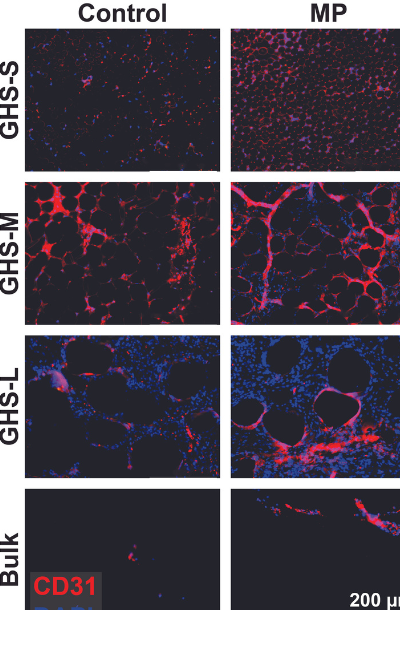
At one week, immunofluorescence staining showed that the hybrid approach resulted in a more significant increase in the migration and accumulation of cells within tissues (cellular infiltration) compared with the other study groups.
The researchers also measured the blood flow (perfusion) at one week by injecting dye into the rats’ blood vessels and using artificial intelligence for detection and measurement. Having identified the granular scaffold made up of medium-sized microgel as optimal for revascularization, the researchers wanted to determine whether adding micropuncture made a difference. The researchers observed more perfusion and longer blood vessel length and, on average, larger vascular density, and vessel diameter, in the group of rats that received the hybrid approach than the group that received only the scaffolds.
“The perfusion in patterned blood vessels indicates that the new blood vessel network is functional, interconnected, and guided by the microarchitecture of granular hydrogel scaffolds. That represents a significant advancement in tissue engineering prior to clinical translation,” said Sheikhi.
The researchers plan to continue investigating their hybrid approach in small animals to better understand the processes, and then conduct research in large animals, and eventually in people.
The study was funded in part by NIBIB (R56EB032672).
This science highlight describes a basic research finding. Basic research increases our understanding of human behavior and biology, which is foundational to advancing new and better ways to prevent, diagnose, and treat disease. Science is an unpredictable and incremental process—each research advance builds on past discoveries, often in unexpected ways. Most clinical advances would not be possible without the knowledge of fundamental basic research.
Study Reference: Zaman Ataie et al. Accelerating Patterned Vascularization Using Granular Hydrogel Scaffolds and Surgical Micropuncture. Small (2023). DOI: /10.1002/smll.202307928
About the graphics: They are both licensed under the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license.

